
The sideways overlap of p orbitals results in a larger internuclear distance between the bonding atoms, leading to longer bond lengths compared to sigma bonds. Bond Length: Pi bonds are typically longer than sigma bonds.Consequently, pi bonds are more susceptible to breaking under the influence of external forces. The overlapping of p orbitals is not as effective in maintaining a strong bond as the head-on overlap of s orbitals in sigma bonds. Bond Strength: Pi bonds are generally weaker than sigma (σ) bonds.The overlapping occurs above and below the plane of the bonded atoms, resulting in a cloud of electron density. Electron Distribution: Pi bonds are formed by the sideways overlap of p orbitals that are parallel to each other.It is characterized by the following properties: Characteristics of Pi BondĪ π (pi) bond is a type of covalent bond formed by the overlapping of p orbitals between atoms. Pi bonds are named for the Greek letter pi, which is used to represent the angle between the two atoms involved in the bond. These bonds are weaker than sigma bonds, but they are still relatively stable. So just remember, as we increase the strength, the covering for the Sigma Bond, we have to compromise on the length of the bond itself.Pi bonds are covalent bonds that occur when two atoms share two pairs of electrons. 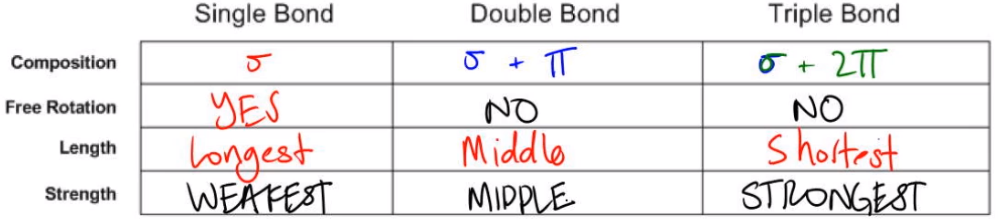
Are single bond are Sigma Bond is very protected and insulated because we have a pi bon on the top in the bottom. But look at the length the length of a little bit shorter and then when we get to the triple bond, were very protected. Its strength of this increasing because we added a pie bond. So if we look yes, we can see here that the single bond we see that it's the longest, and then we see a double bond. So we have here on top and here on the bottom, so this Sigma Bond in the middle is super protected. So here a Sigma bond has triple bond has one Pi Sigma Bond and two Pi bonds. Each pie bond, it's added, makes that bond makes the total bond a little bit stronger. So no matter if you're looking at a single bond, a double bond or triple bond, it's always one Sigma Bond. When looking at a Sigma Bond versus a pie bond, the Pi Bon is just offering, offering a little bit of cover or protection for the Sigma Bond, Not what triple Bond still has only one Sigma bond. It's just offering a little bit more protection for your phone itself. By itself, the phone cover is not stronger than your phone. Think of that as your Sigma Bond, the phone cover that you have on it. Phone your cellphone itself the solid piece of metal or plastic in your hand. 
Now, individually, that pie bond is not stronger than a Sigma Bond is just adding a little bit of extra cushion. So it has one pie bond, which is this red bond here. But then, remember, we set up I bond insulates and protects the Sigma bond. Ah, double bond has still one Sigma bond, which is still this bond here. Now, if we look at a single bond, ah, single bond has one Sigma bond, which remember, is the strongest type of on which directly connects to elements together, and it has zero pi bonds, so here to carbon single body to each other, we can see the length of the bond involved when we talk about a double bond. So let's talk about this whole idea of Sigma Pi bonds when looking at single double and triple bonds. And what we need to realize here is that as the number off pi bonds increases between elements, the bond strength increases, but the bond length decreases. Ah, Pie Bond, which uses the simple pine, is the weaker form of a Kobelev bond that insulate and protects the Sigma bond. It's the strongest form of a Kobelev bond that directly connects elements together. Now, with these types of chemical bonds, we can talk about sigma versus pie bonds now a Sigma bond, which uses the symbol Sigma. And that's why a chemical bond a single chemical bond has to valence electrons involved. That single bond Each one contributes an electron to do it, so they're each contributing a valence electrons. So let's say you have Element A and Element a the bond that connects them. A single Covalin bond involves the sharing of to valence electrons between elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
